weak/strong • concentrated/diluted
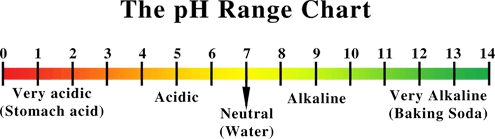
When talking about acids and alkalis, we use terms such as
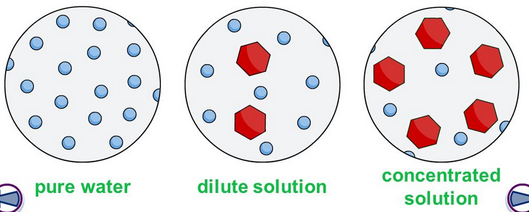
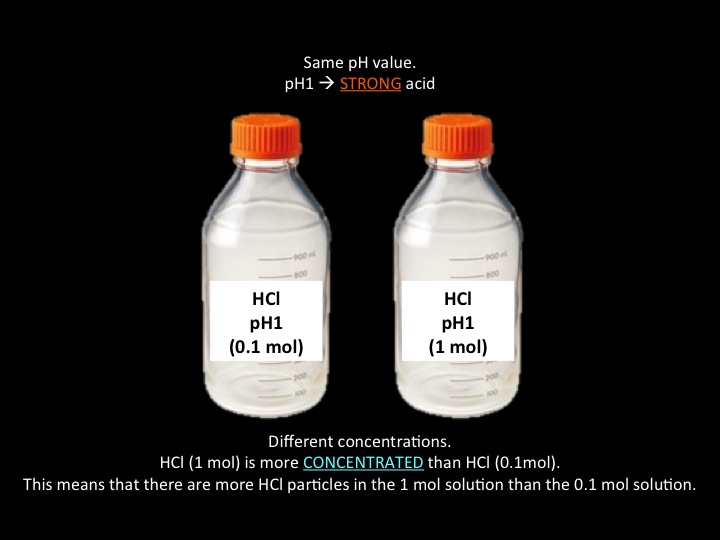
WEAK, STRONG, CONCENTRATED, and DILUTED.
Take note that all these terms have different definitions and MUST NOT BE USED INTERCHANGEABLY.
WEAK, STRONG, CONCENTRATED, and DILUTED.
Take note that all these terms have different definitions and MUST NOT BE USED INTERCHANGEABLY.
So, how does the difference in concentration affect the rate (how fast or how slow) of a chemical reaction?
Watch this amazing video!